Discover your healthcare market with Mursmedic Group. Learn More
Thailand
Demography
Economy

Politics
SYSTEM

Presidential Republic
Indonesia operates as a unitary republic with a presidential system, with power centralized but administrative authority decentralised to regions
SYSTEM

Pro-investment, decentralised governance with strong executive leadership
SYSTEM

Expanding healthcare system and regional autonomy affect market entry

RECENT SIGNAL
Strengthened U.S. trade and investment ties under current administration
Source:
Encyclopaedia Britannica, World Health Organization, Reuters, Indonesian Ministry of Trade (2026)
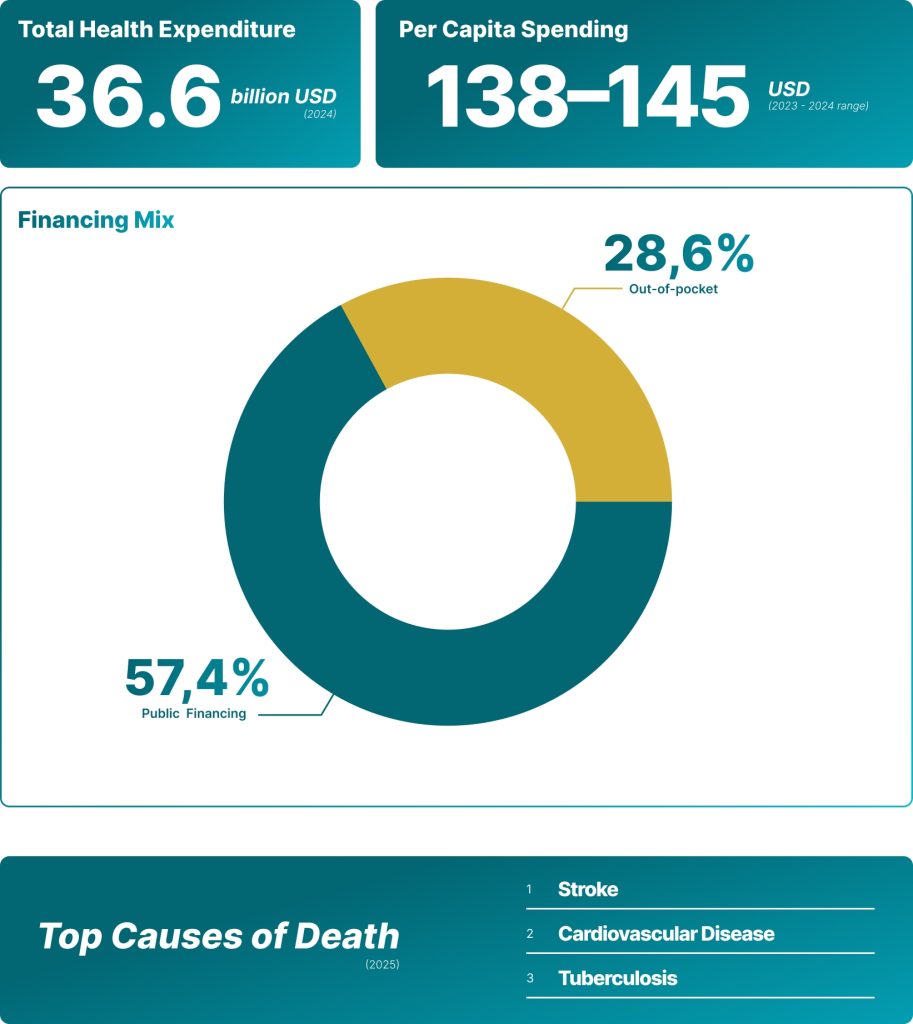
Health

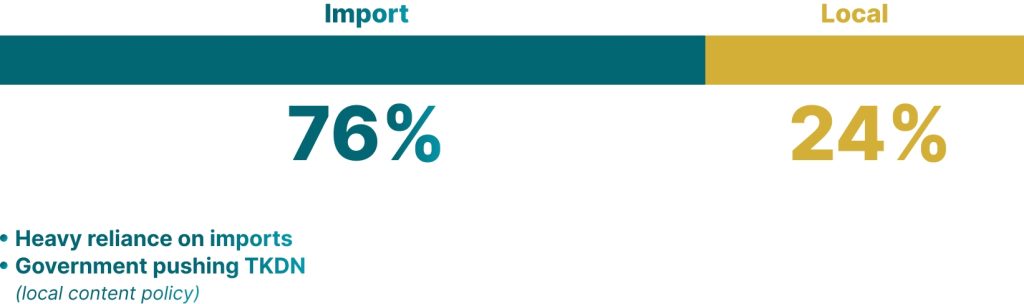
Product Coverage
- Medical Device
- Household Healthcare Product

Product Coverage
- Food
- Drinks
- Cosmetics
- Medicine
Market Entry
In Indonesia, channel choice determines pricing power, compliance burden,
and level of control over your product lifecycle.
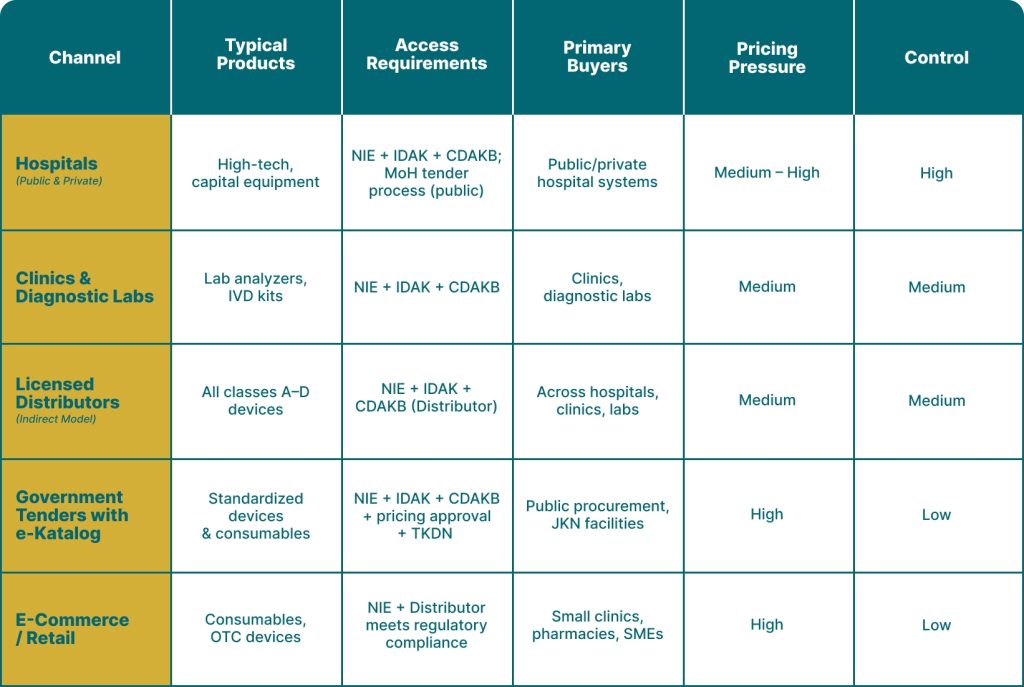
Source:
Kementerian Kesehatan Republik Indonesia, Badan Pengawas Obat dan Makanan (BPOM) Republik Indonesia, International Trade Administration
ALTERNATIVE 1
Set up Local Entity ⟶
Becoming License Holder Oneself
PROS
- Large initial costs and investment
- Local labour and talent challenges
- Local regulation complexity
- Must handle Registration,Importation, and Distribution by one self.
CONS
- Large initial costs and investment
- Local labour and talent challenges
- Local regulation complexity
- Must handle Registration,Importation, and Distribution by one self.
ALTERNATIVE 2
Appoint Independent License Holder
PROS
- Simplified Market Access (facilities are regulatory approved to import)
- Lower initial investment (No local entity required)
- Local expertise and minimal liability for high regulatory compliance.
- Appointing multiple “Sub-distributors” for large market reach to end customers
- Product pricing control
- License ownership is transferable to Principal and other Distributors.
CONS
- Independent LH will not be involved in the sales and commercialization activities
ALTERNATIVE 3
Appoint Distributor
PROS
- Established Market Access (extensive network of potential clients and local market)
- Lower initial investment (no local entity required)
- Local regulatory expertise
CONS
- Limited Control of Operation (brand management, product positioning, customer relationships, distribution, importation, etc.).
- Higher profit margin trade off and lower pricing control
- Conflict of interest with competitor products
- License ownership, difficulty in switching distributors or transferring to oneself.
ALTERNATIVE 4
Appoint Independent License Holder
+ Set up Local Entity
PROS
- Phased Investment Approach with lower initial or upfront costs
- Risk mitigation for market performance evaluation and strategy
- Faster market entry with quick regulatory compliance and ample time to create sales channels within the Territory
- No Conflict of Interest in License ownerships and transferability to Principal.
CONS
- Timeline Considerations: Slight vacuum of business period, where no new importation and sales of product is allowed during transition phase.
- Large initial costs and investment (entity and staffs)